How does the action of millions of molecular motors enable muscle, nature’s most versatile material, to power movement?
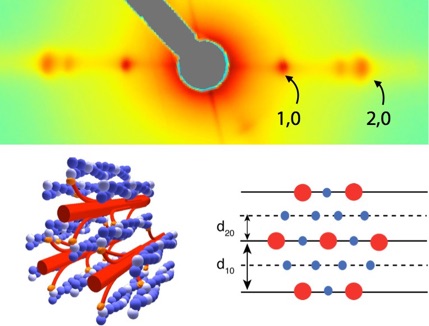
Actin (blue) and Myosin (red) form the contractile lattice that underlies muscle’s motility (lower left). Spacing between filaments (d10 and d20 -- lower right) can change as the muscle is strained. Small-angle x-ray diffraction allows us to image the spacing and mass distribution between the filaments (upper -- grey area is a non-imaged region). Spacing of the 1,0 and 2,0 spots allow us to estimate d10 and d20 and their relative intensity indicates the proportion of myosin heads bound to actin filaments.
One of the features that makes muscle so unique is the exquisite organization of millions of molecular motors into a highly regular protein lattice. Muscles contract via the interaction of many parallel protein fibers (actin & myosin) that form lateral connections (crossbridges). Each crossbridge is a molecular motor and their combined action slides the filaments over one another. While we have a growing understanding of the molecular dynamics of individual motor proteins and of a muscle’s basic material properties, we know very little about the dynamic properties of muscle, especially under perturbed and periodic conditions. Moreover, we would like to understand how these macroscopic properties arise from the statistical mechanics of the millions of molecular repeats. Fortunately, muscle’s microscopic structure is so regular that a highly columnated (small angle) x-ray source directed through the muscle will create a highly resolved diffraction pattern. This can even be accomplished while the muscle is contracting in an animal. Moreover as muscle contracts, the diffraction patterns change. This gives us a window (at up to 2000 frames per second) into the molecular dynamics at the same time we are measuring the macroscopic material properties of the system. In collaboration with Tom Irving (BioCAT director and Professor of biophysics at IIT), we are exploring the dynamics of cross-bridge binding during periodic muscle contraction and their dependence on temperature and the phase of neuromuscular activation. With Dr. José Alvarado and Professor Peko Hosoi at MIT as well as Dr. Dave Williams and Professor Tom Daniel at the University of Washington, we are investigating the lattice arrangement, spacing changes, and hierarchical structure of muscle at the mesoscale. We are connecting the molecular action of myosin binding to the versatile material properties of muscle operating under physiological conditions.